Circular Dichroism Spectrophotometer
J-815 CD, Jasco

In circular dichroism (CD) spectroscopy, the absorbance of right and left circularly polarized light by molecules in solution is measured. Optically active, chiral molecules absorb one direction of circular polarized light over the other. This differential absorption provides information on molecular structure and its changes for small molecules, proteins and nucleic acids. For example, changes in secondary structure caused by protein unfolding (induced by perturbants such as temperature or pH), or folding (induced by the binding of a small molecule ligand, protein, or nucleic acid) can be quickly and easily quantified as changes in the far-UV CD spectrum. Separately, CD spectroscopy is also an essential technique in the study of chiral-selective properties of all molecules, including drugs, fluorescent dyes, and chemical catalysts designed to mimic enzymatic catalysis.
Features
- Scanning emission monochromator for fluorescence measurements
- Bio-Logic SFM 4 syringe stopped-flow system: 2-4 solutions can be mixed and injected into a cuvette with a dead time < 1 ms
- Pelitier temperature control from 4 - 110°C
- Multiple acquisition modes:
- Wavescan from 163-900 nm
- Temperature scan, which collects a full wavescan or data at up to 8 wavelengths at every indicated temperature interval
- Fixed wavelength time scan for chemical denaturation and stopped-flow experiments
-
Scan speeds of up to 10,000 nm/min allows rapid data collection
-
Low noise (RMS noise < 0.02 mdeg at 200 nm) and very low stray light (< 0.0003% at 200 nm)
-
Non-destructive technique
- 1 cm, 1 mm and 0.1 mm cuvettes available
Applications
- Protein folding and conformation studies:
- Estimate protein secondary structure composition
- Measure changes produced upon protein folding, ligand binding, or mutation
- Measure changes in stability in response to environmental perturbations
- Purity testing of optically active substances
- DNA/RNA interactions and conformation
- Drug development:
- Determine stereochemistry of chiral drugs and proteins
- Monitor ligand-induced protein or peptide conformational changes
- Gather information on binding sites
- Enzyme kinetics
Internal Rates
- Unassisted: $33.00/hr
- Assisted: $84.00/hr
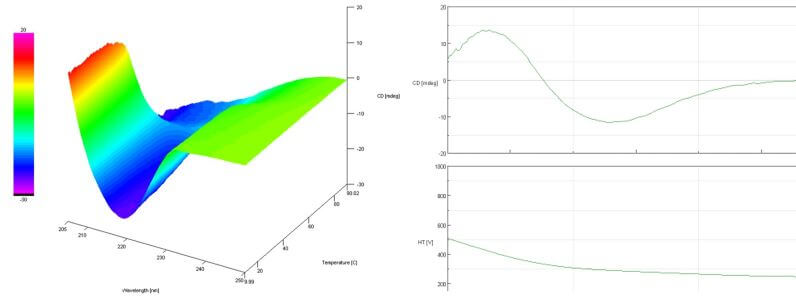
Left: Example temperature scan of a protein. Right: Wavescan of a β-helix protein.